The Profit Motive Behind New Alzheimer’s Treatments
Big Pharma and industry-funded advocacy groups are promoting Alzheimer’s blood testing that could label millions of Americans as sick — despite dangerous treatments for early diagnoses of the disease.
Big Pharma and allied advocacy groups are pushing early Alzheimer’s screening tests that could make millions eligible for risky drugs. (Paul Bersebach / MediaNews Group / Orange County Register via Getty Images)
When Barbara Feuerstein, seventy-two, saw a Facebook ad offering a free blood test for Alzheimer’s disease, she jumped at the opportunity. The ad, placed by a research center, offered the test, recently cleared by the Food and Drug Administration (FDA), as part of a drug study to prevent the disease. “I figured it must be safe if it was FDA approved,” said Feuerstein.
It surely helped that prominent experts had launched an impressive campaign to convince doctors, the public, and legislators to embrace widespread Alzheimer’s testing. In impassioned op-eds published around the country, Robert Redfield, former director of the Centers for Disease Control and Prevention (CDC), proclaimed, “These tests fundamentally change our understanding of the disease, just as the HIV test did for AIDS.” At senior communities and other locales across the country, mobile testing vans were offering free blood checks just outside people’s doors.
And the Alzheimer’s Association, a private institution that funds Alzheimer’s research and whose recommendations on the disease are often taken as the final word, has been promoting bipartisan legislation to ensure the test will be covered by Medicare as a screening mechanism for millions of individuals without any memory problems.
But what Feuerstein and millions of others didn’t realize was that many of the tests’ biggest advocates are being paid millions by the drug and device companies that stand to gain from people taking these tests and opting for preventive drug treatment. These same companies are pushing the Medicare legislation that could radically expand their business.
If such efforts are successful, sales of the new blood tests and drugs could explode. Currently, only 592,000 people are eligible for treatment with the new Alzheimer’s drugs. But if the pharmaceuticals are used to prevent rather than treat the disease, the market pool jumps to forty-seven million Americans.
The campaign is already paying off. A 2025 survey by the Alzheimer’s Association found that more than nine out of ten people would like to take the Alzheimer’s blood test so they could start early treatment. Many are already getting tested, either by doctors who order the exams “off label” or by purchasing at-home tests, no prescription necessary. (And while Marty Makary, the FDA commissioner who oversaw the clearance of the new tests, has now been ousted, there’s little reason to suspect his replacement will be any less industry friendly.)
It’s true that the new blood tests are “highly accurate” in identifying a biological marker associated with Alzheimer’s in people already suffering from dementia. But when they’re used to screen for Alzheimer’s in people like Feuerstein, who may be a bit forgetful but otherwise cognitively normal, they’re less accurate than flipping a coin.
Feuerstein didn’t realize this when her blood test turned up the biological marker associated with Alzheimer’s, so she agreed to take Kisunla, a “breakthrough” new Alzheimer’s drug the study’s researchers were testing. She wasn’t aware of the drug’s questionable efficacy — and didn’t realize it could cause disabling and fatal side effects. But she’d soon learn that for herself.
At the start of the study, Feuerstein was independent and active. But shortly after her third Kisunla dose, the former bookkeeper became confused and unable to walk without assistance. She couldn’t write her name and had trouble operating her toaster.

A head CT scan showed massive brain swelling — a well-known complication of Kisunla. Following treatment and repeated hospital stays, doctors sent Feuerstein home. Still somewhat confused and struggling with her balance, she walked her dog, Daisy, late one evening and fell in the street, breaking her hip.
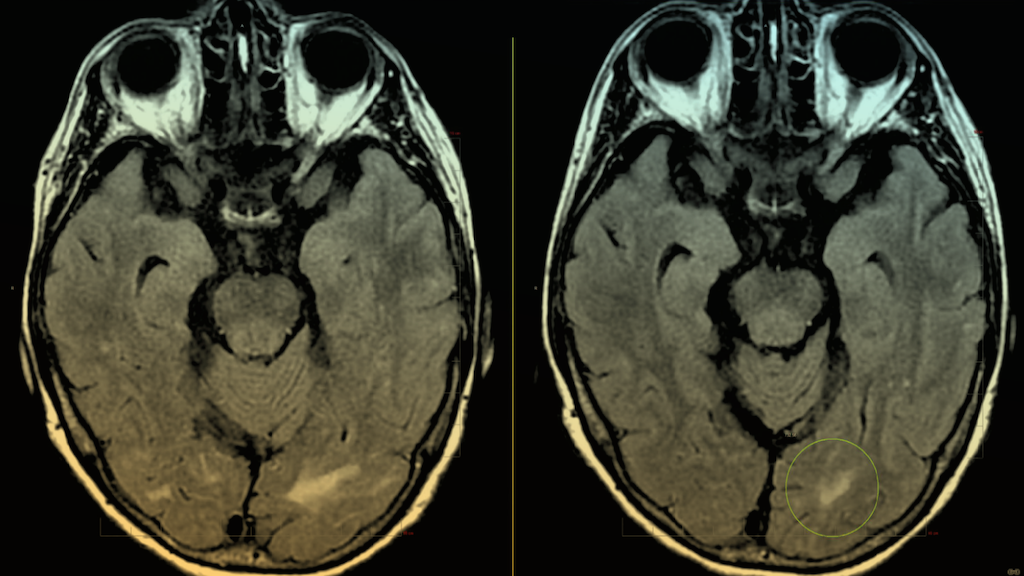
“I screamed for help, but no one could hear me,” she says. “I thought I was going to die.” She lay in the street until 1 a.m., when a passing motorist spotted her.
Feuerstein is far from alone. In the pivotal study of Kisunla, 37 percent of individuals given Kisunla developed brain swelling and hemorrhages. Manufacturers of the new Alzheimer’s drugs say these complications are largely “benign and self-resolving,” a determination often made when patients’ MRI scans return to normal. But patients like Feuerstein and others tell a different story.
Kristine Shields, sixty-five, developed visual hallucinations, brain swelling, microhemorrhages, and seizures after receiving an experimental anti-amyloid drug in the same class as Kisunla. Like Feuerstein, Shields’s recent MRI scans show her brain swelling has resolved, but because of her seizures and brain fog, she lost her job. She also lost her driver’s license for eight months and will likely have to take antiepileptic medicines for life.
Some experts worry that even those who don’t experience symptoms like Shields’s and Feuerstein’s could still be harmed by such Alzheimer’s treatments. Along with sometimes triggering brain hemorrhages and swelling, the drugs are also known to cause brain shrinkage. And government researchers have noted that neurofilament light, a protein that builds up with brain cell death, increased during a clinical trial of Kisunla.
The idea of giving potentially dangerous drugs to people based on a blood test for a disease they might never develop doesn’t sit well with George Perry, a neuroscientist and editor in chief of the Journal of Alzheimer’s Disease. According to Perry, that’s like “giving chemotherapy to everyone because they might someday develop cancer.”
Disease Creep and the Damage Done
Until recently, Alzheimer’s was only diagnosed in people with memory or cognitive problems. But in 2011, the Alzheimer’s Association issued a controversial statement that Alzheimer’s was a biological disease primarily caused by the buildup of a protein in the brain called amyloid beta, regardless of any symptoms. The new criteria were developed to align Alzheimer’s with other diseases, such as cancer, which are based on physical findings.
This announcement opened the door to the newly minted diagnosis of “preclinical” Alzheimer’s, an early phase of the disease marked by the presence of amyloid in a patient’s brain or blood without any other symptoms — which, according to the association, can last two decades or longer.
Such “disease creep,” in which the criteria for diagnosing a disease or pre-disease are loosened, is becoming increasingly common for conditions ranging from high blood pressure to diabetes. Critics suspect a profit motive behind the phenomenon: the more people diagnosed with a disease or pre-disease, the larger the market for potential treatment.
If Alzheimer’s were a biological disease tied to the presence of amyloid, that meant doctors could theoretically test for it — if they could find a relatively cheap and easy way to do so. Hope for that seemed to appear in August 2025, when Lumipulse, a Japanese-developed blood test for blood proteins associated with amyloid plaques in the brain, hit the market three months after being cleared by the FDA. Two months later, regulators cleared a similar test, Elecsys, developed by Roche and Eli Lilly.
Such tests are undeniably adept at detecting amyloid; Lumipulse was found to be 89 to 98 percent accurate at identifying such buildups in people’s brains.
That’s promising — except for the fact that the presence of amyloid in the brain turns out to be a terrible predictor of dementia or Alzheimer’s in people who don’t already have other symptoms of the disease.
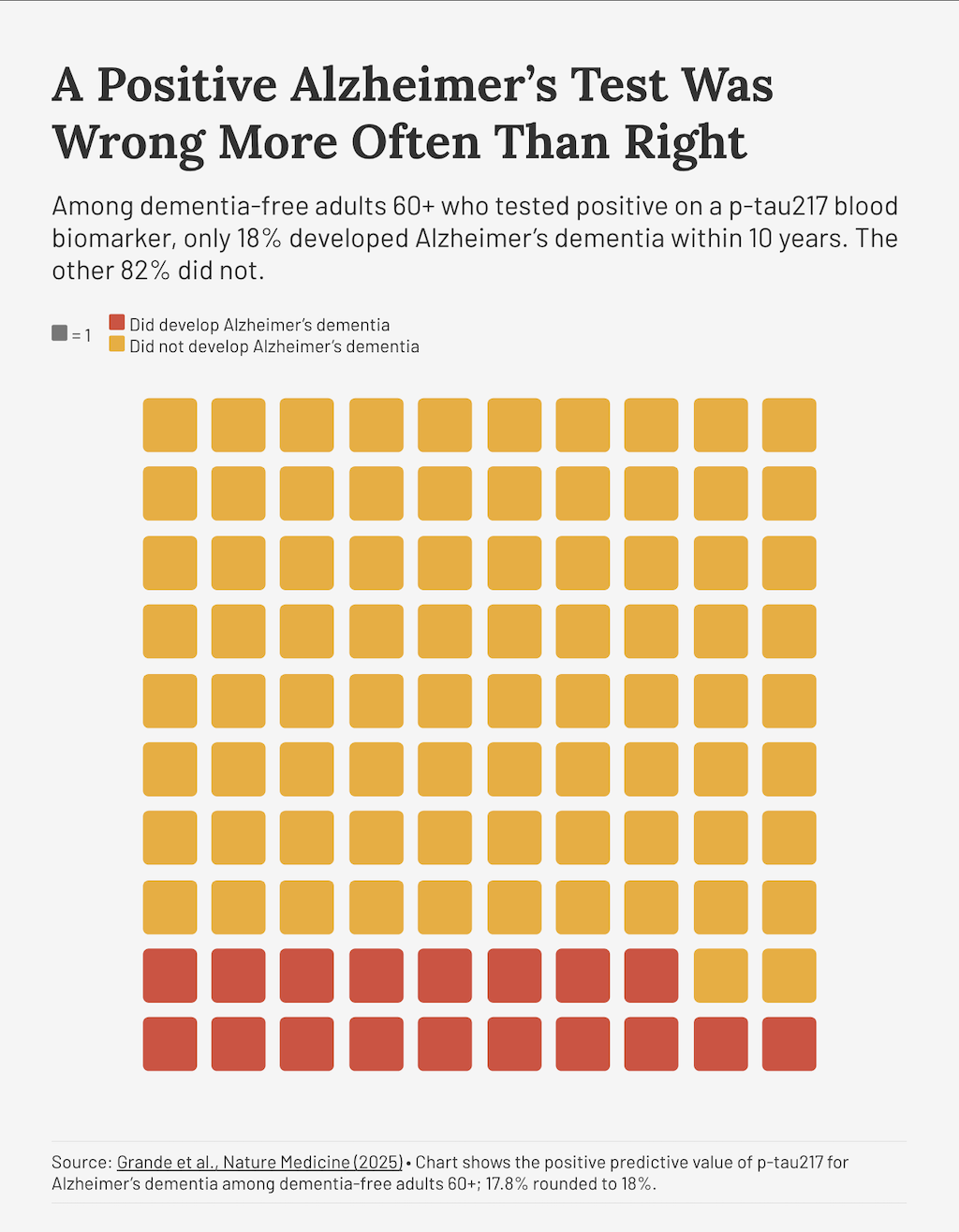
Only a few studies have examined the clinical accuracy of the Alzheimer’s test when used as a screening tool, and the results are dismal. Among cognitively normal people sixty years and older, only 18 percent of those with a positive blood test developed dementia during a ten-year follow-up period — meaning that 82 percent could be falsely labeled with a dreaded disease.
Another study found that although more than a third of adults seventy years and older had amyloid in their systems, only one-tenth had dementia. Even when researchers used a sophisticated combination of blood tests, they were only able to accurately identify people with Alzheimer’s 43 percent of the time.
In the end, most individuals who are cognitively normal and test positive on an Alzheimer’s blood test will never develop Alzheimer’s dementia.
Just as amyloid is a poor predictor of dementia, reducing amyloid with drugs like Kisunla doesn’t necessarily correlate with the slowing of dementia. Researchers at Stanford University reviewed data from the FDA and found that although the drugs effectively removed amyloid, there was “little to no” correlation between the amount of amyloid removed and any benefit.
It’s why a 2024 Lancet standing commission report on dementia, which was not funded by industry, concluded that biological changes alone aren’t sufficient to diagnose Alzheimer’s. And in March 2026, the American College of Physicians and the Annals of Internal Medicine convened a panel of independent experts on the matter and determined that there is no value to screening people with normal cognition for Alzheimer’s “until effective, safe, and cost-effective therapies that yield meaningful clinical improvements” are available.
The likelihood of Alzheimer’s tests delivering false positives isn’t just misleading and upsetting — such results can have serious social and financial ramifications.
A review of state insurance regulations found that individuals who test positive for preclinical Alzheimer’s could be denied long-term care due to them appearing to have “preexisting conditions.” Another review found positive test results could lead to other forms of discrimination, such as denial of employment, with the Americans with Disabilities Act offering only limited options for protection and recourse.
What’s more, according to the International Working Group, a group of experts that provides updated guidance on the diagnosis of Alzheimer’s, cognitively normal individuals who are positive for a single blood biomarker have a “high chance of never developing cognitive impairment in their lifetime,” and that the resulting “psychological and societal ramifications of being labeled with Alzheimer’s disease [can be] consequential.”
The campaign to diagnose and treat preclinical Alzheimer’s is unfortunate, says Gayatri Devi, a neurologist and clinical professor of neurology at Zucker School of Medicine at Hofstra University. She worries that diagnosing individuals with preclinical Alzheimer’s will cause “enormous anxiety” for many people who will think that the term means it’s just a matter of time before they develop dementia.
Devi, who does not receive funding from the pharmaceutical industry, says, “The criteria’s simplicity appeals to both time-strapped physicians and pharmaceutical companies eager to expand treatment access.”
Colleen Christmas, a geriatrician and chair-elect of the American College of Physicians, who also does not receive industry support, says the correlation between biomarker findings and Alzheimer’s is “loose.”
“So,” she asks, “why are we doing the blood tests, other than to make people terrified and ask for the drugs?”
Money Shots
In a syndicated op-ed published in newspapers across the country, including the Chicago Tribune and the Seattle Times, Redfield, the former CDC director, has been sounding the call for people to take Alzheimer’s tests.
According to Redfield, “Alzheimer’s begins when amyloid becomes abnormal in the brain, decades before symptoms arise.” But nowhere in these pieces was it disclosed that from 2021 to 2024, Redfield was paid $638,387 for consulting services by Roche Diagnostics, a manufacturer of one of the new Alzheimer’s tests.
Redfield did not respond to multiple emails and calls requesting comment.
Similarly, many of the mobile vans that offer free testing around the country are run by companies called contract research organizations, which are paid by drugmakers to conduct clinical trials of their drugs. Such free testing is a way to recruit subjects for their studies.
That includes Artemis, the contract research organization that tested Feuerstein, which was paid by Eli Lilly, the manufacturer of Kisunla, the drug Feuerstein was prescribed. Artemis refused to grant an interview or answer questions for this story, stating that they have a policy against speaking with the media. Lilly declined to respond to questions about how much it pays centers to enroll volunteers.
And the Alzheimer’s Association, which currently champions both the Alzheimer’s blood tests and corresponding Alzheimer’s drugs for individuals with cognitive loss, receives millions in funding from companies that stand to gain from such promotion. In 2025, the organization received more than $1 million each from Eisai and Eli Lilly, the manufacturers of Leqembi and Kisunla, respectively; $875,000 from Biogen, the co-manufacturer of Leqembi; more than $819,000 from Novo Nordisk, which is also developing drugs to treat Alzheimer’s, and $315,000 from Genentech/Roche, codeveloper with Lilly of the new Elecsys Alzheimer’s blood test.
Maria Carrillo, chief science officer of the Alzheimer’s Association, told the Lever that the Alzheimer’s blood tests should only be used in “patients with cognitive impairment being seen in specialized care for memory disorders.” But the association is already paving the way to expand testing and treatment to people who are cognitively normal.
It’s doing so by supporting the Alzheimer’s Screening and Prevention Act of 2025, known as the “ASAP Act,” which “creates a pathway” to mandate Medicare payments for “blood-based dementia screening tests.” In 2025, the association’s lobbying arm, the Alzheimer’s Impact Movement, spent nearly $4 million and retained twenty-one lobbyists to lobby for issues including the ASAP Act, which the lobbying operation called a “mammogram moment” for Alzheimer’s.
According to Carrillo, “The bill has more than fifty cosponsors in the House and eighteen in the Senate — bipartisan support that reflects the urgency this moment demands.” She added that the association supports the bill because “under current law, Medicare is prohibited from covering screening tests unless Congress specifically permits it or the US Preventive Services Task Force recommends it — a process that can take up to seven years.”
The House ASAP Act is cosponsored by Reps. Paul Tonko (D-NY) and Vern Buchanan (R-FL) — and both have financial ties to Alzheimer test companies and their surrogates.
In the 2024 election cycle, Tonko received $10,000 from the Alzheimer’s Association and another $234,000 from other health-sector interests, his second-largest industry funders, according to OpenSecrets. In an email, Tonko’s communications director noted that the new Alzheimer’s blood tests, “while not a panacea, can be a valuable tool in the toolbox for providers as they work to address this devastating disease, and Medicare patients deserve access to these advances.”
The health sector was Buchanan’s top 2024 industry funder, donating $513,000 to his campaign, according to OpenSecrets. That included $10,000 from Abbott Laboratories, which produces an Alzheimer’s blood test; and $15,000 from Amgen, which developed umibecestat, an experimental Alzheimer’s drug; In addition, he received $5,000 from the Alzheimer’s Association.
The Alzheimer’s Association isn’t the only industry-funded advocacy organization lobbying politicians to pass the bill; such practices have become increasingly common as drug companies have worked to influence the country’s pharmaceutical pipeline.
The Alliance for Aging Research, a large nonprofit that promotes “healthy aging and equitable access to care,” spent $437,000 on lobbying during the 2024 election cycle for matters including the ASAP Act, which it calls an “avenue to unprecedented hope for earlier diagnosis [and] treatment of Alzheimer’s.”
The alliance took in $5,360,000 in revenues in 2024, but it does not reveal its funders. Its “Corporate Partnership Council” includes representatives from Biogen, co-manufacturer of the first two new Alzheimer’s drugs, as well as Eli Lilly and the Pharmaceutical Research and Manufacturers of America (PhRMA), the pharmaceutical industry’s main lobbying group. An Alliance for Aging Research spokesperson told the Lever that “the majority” of its funding comes from drug and device makers but declined to reveal the exact percentage.
Bad Tests, Bad Drugs
If efforts to pass the ASAP Act are successful and testing and treatment for preclinical Alzheimer’s becomes widespread, drug and device makers can anticipate a financial windfall. An estimated forty-seven million people in the United States have so-called preclinical Alzheimer’s, based on the Alzheimer’s Association’s criteria. That number dwarfs the less than 600,000 people with early Alzheimer’s who are currently eligible for treatment with anti-amyloid drugs.
A market explosion of this size holds enormous interest for investors. It also holds interest for members of the public who don’t know the country’s history of failed screening tests that have caused serious harm. For example, an ambitious screening program in the 1980s tested infants’ urine for neuroblastoma, a rare childhood cancer, but the effort was abandoned after the program not only failed to reduce deaths but also led to significant harms due to overdiagnosis and treatment.
Given the rising number of people projected to have Alzheimer’s in the coming years, a sense of urgency in developing new treatments is understandable. But Adriane Fugh-Berman, an expert in analyzing drug studies at Georgetown University and codirector of the evidence-based prescription nonprofit organization PharmedOut, says that urgency shouldn’t outweigh scientific evidence.
“Early detection isn’t always a good thing,” she said. “These are bad tests designed to make people eligible for bad drugs.”